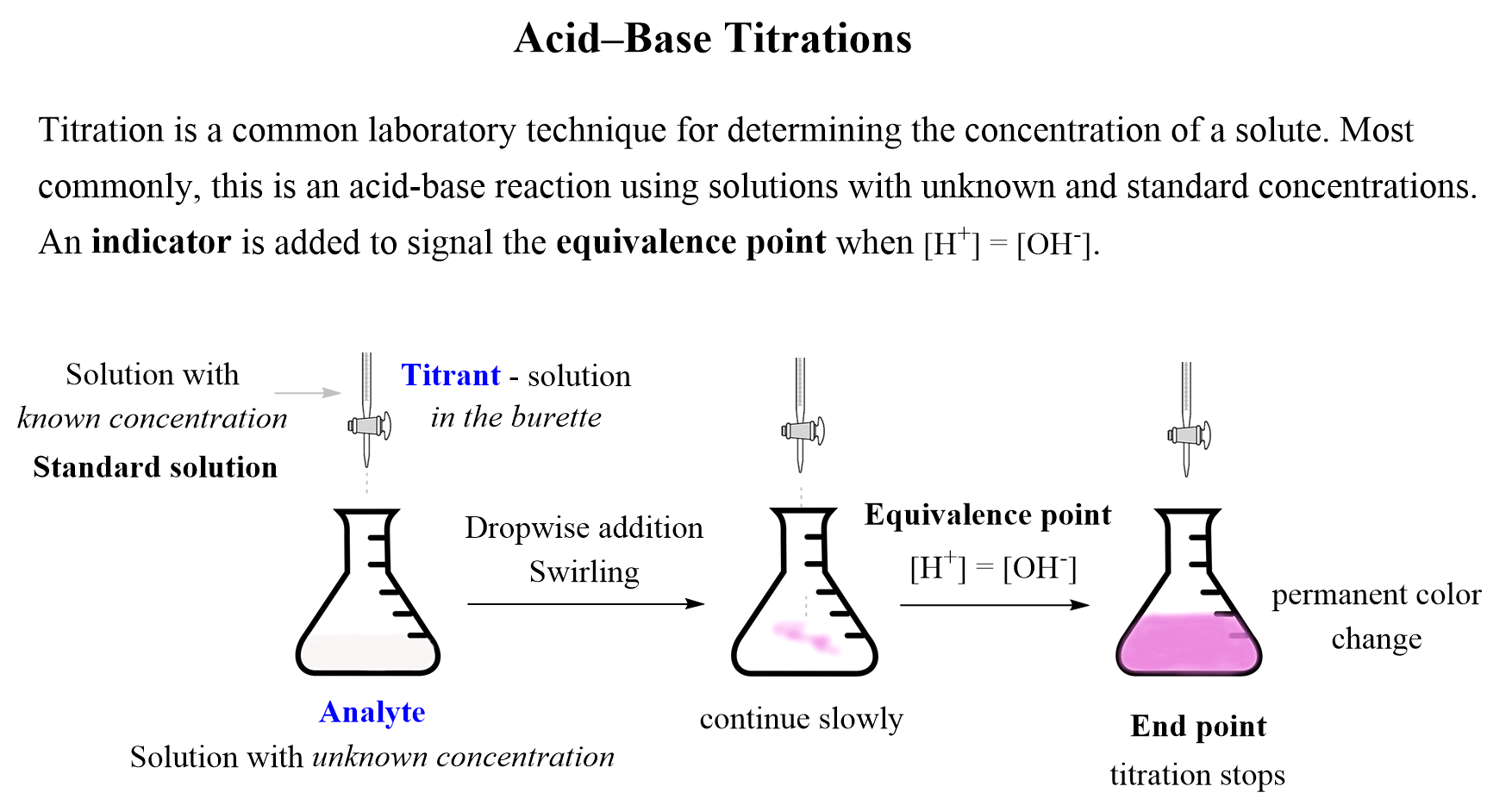
Discussion For Titration Experiment . In a ph titration you measure the ph as a function of the volume of titrant added and determine the equivalence point as the point in. Volumetric procedures are among the most common and convenient.
from general.chemistrysteps.com
In a ph titration you measure the ph as a function of the volume of titrant added and determine the equivalence point as the point in. In a titration, the conclusion is often a simple statement of the experimentally. Volumetric procedures are among the most common and convenient.
AcidBase Titrations Chemistry Steps
Discussion For Titration Experiment In this experiment you will be determining the volume of sodium hydroxide solution of known concentration. Titration is the process of determining the concentration of an unknown solution using a solution of known. write your conclusion. Volumetric procedures are among the most common and convenient.
From letitsnowglobe.co.uk
Titration procedure pdf Discussion For Titration Experiment write your conclusion. In this experiment you will be determining the volume of sodium hydroxide solution of known concentration.a titration is an experiment where a volume of a solution of known concentration is added to a volume of another solution in order to. In a ph titration you measure the ph as a function of the volume. Discussion For Titration Experiment.
From www.microlit.com
An Advanced Guide to Titration Microlit Discussion For Titration Experiment Titration is the process of determining the concentration of an unknown solution using a solution of known. In this experiment you will be determining the volume of sodium hydroxide solution of known concentration. In a ph titration you measure the ph as a function of the volume of titrant added and determine the equivalence point as the point in. Volumetric. Discussion For Titration Experiment.
From mungfali.com
Titration Steps Discussion For Titration Experimentin this experiment students neutralise sodium hydroxide with hydrochloric acid to produce the soluble salt sodium chloride in solution. Titration is the process of determining the concentration of an unknown solution using a solution of known.a titration is an experiment where a volume of a solution of known concentration is added to a volume of another solution. Discussion For Titration Experiment.
From www.studocu.com
Lecture Notes 9 + Experiment 9 ACIDBASE TITRATION Experiment 9 Discussion For Titration Experiment In a ph titration you measure the ph as a function of the volume of titrant added and determine the equivalence point as the point in.a titration is an experiment where a volume of a solution of known concentration is added to a volume of another solution in order to. write your conclusion.in this experiment. Discussion For Titration Experiment.
From www.science-revision.co.uk
Titrations Discussion For Titration Experiment In a titration, the conclusion is often a simple statement of the experimentally.a titration is an experiment where a volume of a solution of known concentration is added to a volume of another solution in order to. In this experiment you will be determining the volume of sodium hydroxide solution of known concentration.a titration is an. Discussion For Titration Experiment.
From letitsnowglobe.co.uk
Titration procedure pdf Discussion For Titration Experiment In a titration, the conclusion is often a simple statement of the experimentally. Titration is the process of determining the concentration of an unknown solution using a solution of known.in this experiment students neutralise sodium hydroxide with hydrochloric acid to produce the soluble salt sodium chloride in solution. In a ph titration you measure the ph as a. Discussion For Titration Experiment.
From www.thinkswap.com
Complete Acid/Base Titration Report Chemistry Year 12 HSC Thinkswap Discussion For Titration Experiment In a titration, the conclusion is often a simple statement of the experimentally. write your conclusion. In this experiment you will be determining the volume of sodium hydroxide solution of known concentration.a titration is an experiment where a volume of a solution of known concentration is added to a volume of another solution in order to. Volumetric. Discussion For Titration Experiment.
From www.chemicals.co.uk
What is Titration in Chemistry? The Chemistry Blog Discussion For Titration Experiment In this experiment you will be determining the volume of sodium hydroxide solution of known concentration. write your conclusion. In a titration, the conclusion is often a simple statement of the experimentally. In a ph titration you measure the ph as a function of the volume of titrant added and determine the equivalence point as the point in. Web. Discussion For Titration Experiment.
From letitsnowglobe.co.uk
Titration procedure pdf Discussion For Titration Experiment Titration is the process of determining the concentration of an unknown solution using a solution of known. In a ph titration you measure the ph as a function of the volume of titrant added and determine the equivalence point as the point in.a titration is an experiment where a volume of a solution of known concentration is added. Discussion For Titration Experiment.
From www.chemicals.co.uk
How To Carry Out a Titration Experiment The Chemistry Blog Discussion For Titration Experimenta titration is an experiment where a volume of a solution of known concentration is added to a volume of another solution in order to. write your conclusion. Titration is the process of determining the concentration of an unknown solution using a solution of known. Volumetric procedures are among the most common and convenient.in this experiment. Discussion For Titration Experiment.
From www.youtube.com
Titration of Oxalic Acid vs NaOH oxalic acid vs NaOH titration Discussion For Titration Experiment Titration is the process of determining the concentration of an unknown solution using a solution of known. In a ph titration you measure the ph as a function of the volume of titrant added and determine the equivalence point as the point in. In this experiment you will be determining the volume of sodium hydroxide solution of known concentration. Web. Discussion For Titration Experiment.
From www.scribd.com
titration ( chemistry Experiment report) Titration Analysis Discussion For Titration Experiment In this experiment you will be determining the volume of sodium hydroxide solution of known concentration.a titration is an experiment where a volume of a solution of known concentration is added to a volume of another solution in order to. Titration is the process of determining the concentration of an unknown solution using a solution of known. In. Discussion For Titration Experiment.
From fity.club
Titration Experiment Discussion For Titration Experimentin this experiment students neutralise sodium hydroxide with hydrochloric acid to produce the soluble salt sodium chloride in solution. write your conclusion.a titration is an experiment where a volume of a solution of known concentration is added to a volume of another solution in order to.a titration is an experiment where a volume of. Discussion For Titration Experiment.
From www.priyamstudycentre.com
Acid Base Titration Principle, Types, Process, Indicators Discussion For Titration Experiment Titration is the process of determining the concentration of an unknown solution using a solution of known. Volumetric procedures are among the most common and convenient. In a titration, the conclusion is often a simple statement of the experimentally. write your conclusion. In a ph titration you measure the ph as a function of the volume of titrant added. Discussion For Titration Experiment.
From igcse-chemistry-edexcel.blogspot.com
IGCSE Chemistry 4.9 describe experiments to carry out acidalkali Discussion For Titration Experiment write your conclusion.a titration is an experiment where a volume of a solution of known concentration is added to a volume of another solution in order to. Volumetric procedures are among the most common and convenient.in this experiment students neutralise sodium hydroxide with hydrochloric acid to produce the soluble salt sodium chloride in solution. In. Discussion For Titration Experiment.
From brogan-blogmckenzie.blogspot.com
Acid Base Titration Experiment Report Discussion For Titration Experiment In a titration, the conclusion is often a simple statement of the experimentally. Titration is the process of determining the concentration of an unknown solution using a solution of known. In a ph titration you measure the ph as a function of the volume of titrant added and determine the equivalence point as the point in.a titration is. Discussion For Titration Experiment.
From revisechemistry.uk
Titration OCR GCSE PAG 6 revisechemistry.uk Discussion For Titration Experiment In a titration, the conclusion is often a simple statement of the experimentally. Volumetric procedures are among the most common and convenient.in this experiment students neutralise sodium hydroxide with hydrochloric acid to produce the soluble salt sodium chloride in solution. Titration is the process of determining the concentration of an unknown solution using a solution of known. Web. Discussion For Titration Experiment.
From chemistry.analia-sanchez.net
Titration Notes Chemistry Classes / Ronald Reagan S.H.S. Discussion For Titration Experimentin this experiment students neutralise sodium hydroxide with hydrochloric acid to produce the soluble salt sodium chloride in solution.a titration is an experiment where a volume of a solution of known concentration is added to a volume of another solution in order to. write your conclusion. Volumetric procedures are among the most common and convenient. In. Discussion For Titration Experiment.